 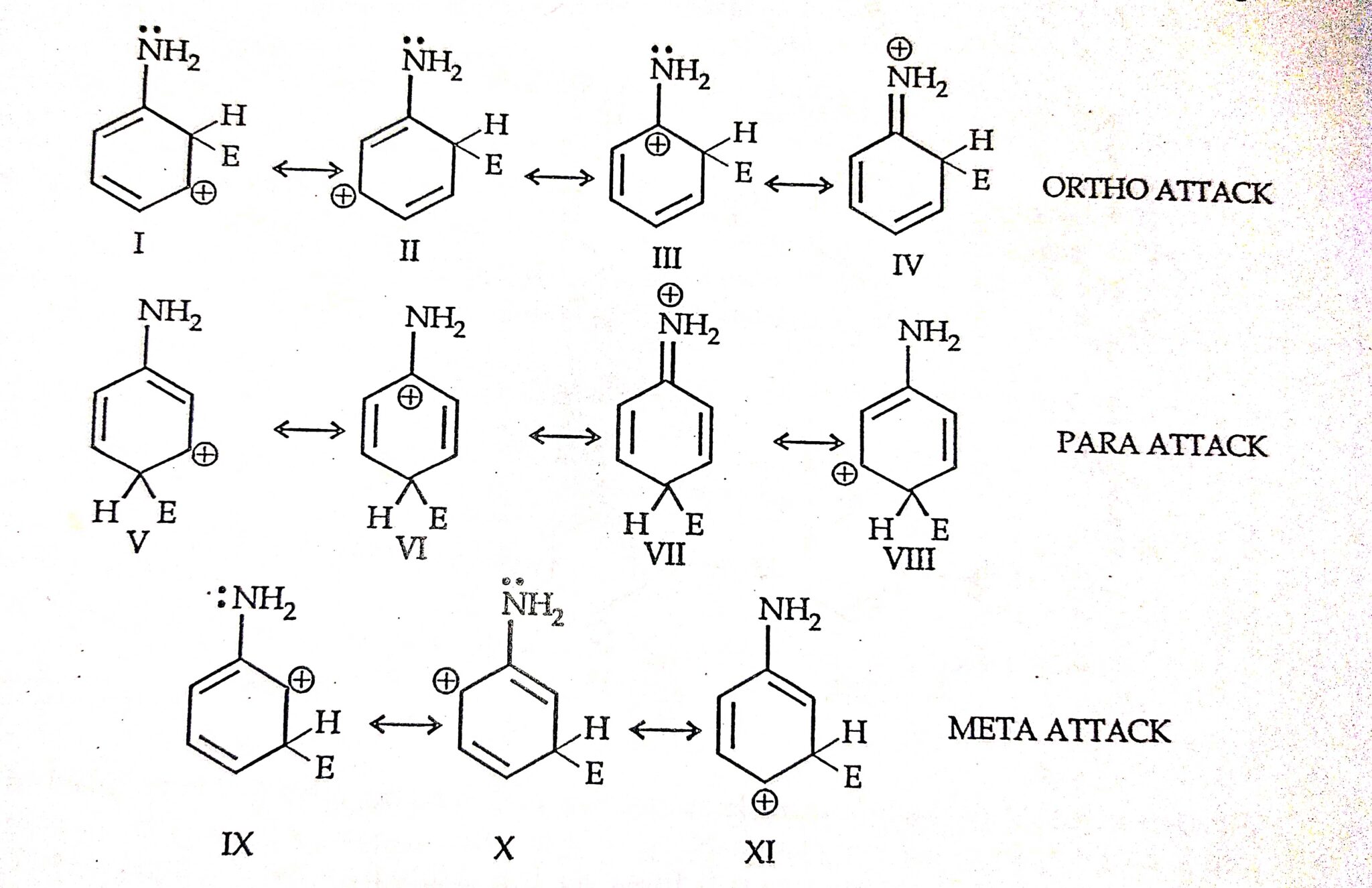
Para represents a 1,4 relationship, or groups that are opposite each other on the benzene ring. Meta represents a 1,3 relationship, or groups that are separated by one carbon on the benzene ring. Ortho represents a 1,2 relationship, or 2 groups that are directly near each other on the benzene ring. Ortho-dimethylbenzene (ortho-xylene or o-xylene) tells us that the 2 groups are directly next to each other on the ring.Ĭommon Mistake Warning : Despite their similar appearance, these relationships ONLY apply to benzene rings and NOT cyclohexane rings. When it comes to a disubstituted benzene, using the terms ‘ortho, meta and para’ is simply another way to indicate the relationship of the substituents to each other, rather than their overall placement on the ring.įor example, 1,2-dimethylbenzene tells us we have methyl groups both on carbons 1 and 2. Given a 5 carbon chain with a methyl on carbon 4 and alcohol on carbon 2, you get 4-methyl-2-pentanol.īut that wasn’t the only way to indicate the relationships of substituents, was it? Think back to cis and trans or to E and Z. In IUPAC naming, every substituent is designated with a number. Let’s backtrack all the way to nomenclature from your first (second?) chapter in orgo 1. In the context of Electrophilic Aromatic Substitution, understanding the chemistry of substituents will help you figure out where to direct the incoming electrophile on a substituted benzene ring. Ortho, Meta and Para refer to the relationship between substituents on a disubstituted benzene ring. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
